Question :
The group of molecules having identical shape is
Options :
(a) PCI5, IF5, XeO2F2
(b) BF3 , PCI3, XeO3
(c) SF4,XeF4,CCI4
(d) CIF3, XeOF2, XeF3+
JEE Main Chemical Bonding 2016 PYQ with solution
Answer :
Correct answer : option (d)
CIF3 :
The central atom : Chlorine
The electronic configuration of chlorine is [Ne] 3s² 3p⁵
The number of valence electrons present in chlorine is 7
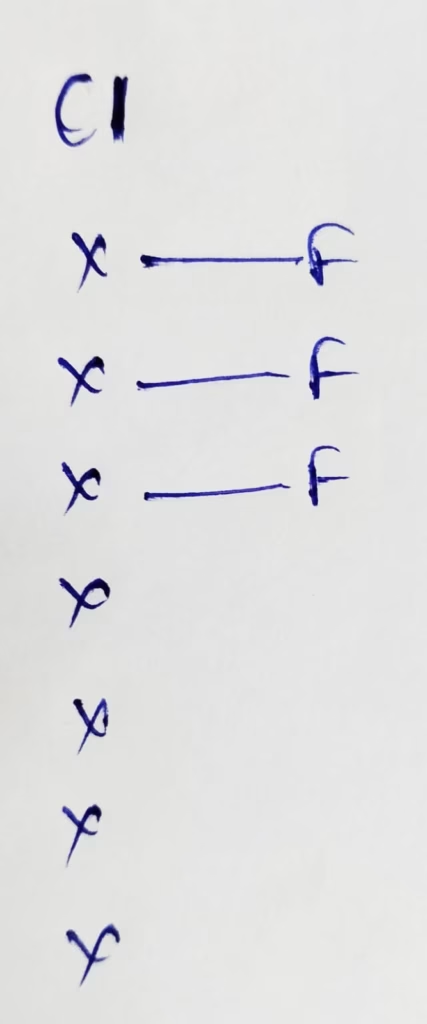
Hybridization : Number of bonds + lone pairs = 3 bonds + 2 lone pairs = 5 { Sp3d hybridization }
The shape of CIF3 molecule is T shape
XeOF2 :
The central atom : Xenon
The electronic configuration of Xenon is [Kr] 4d¹⁰ 5s² 5p⁶
The number of valence electrons in Xenon is 8
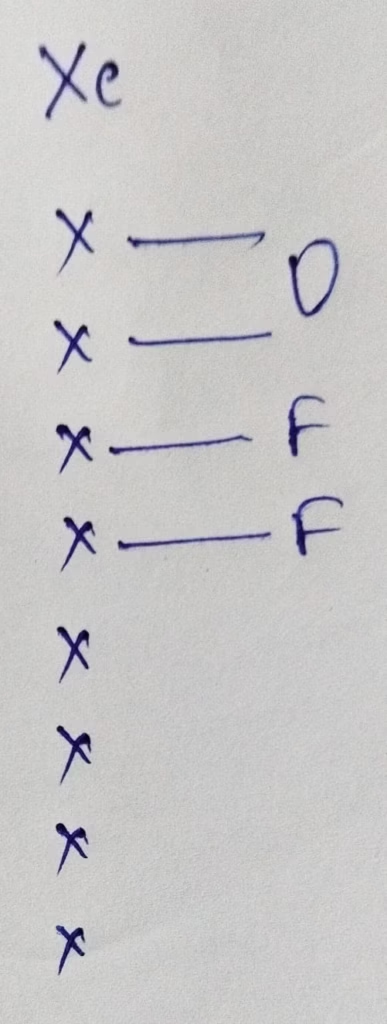
Hybridization : Number of bonds + lone pairs = 3 bonds + 2 lone pairs = 5 { Sp3d hybridization }
The shape of XeOF2 molecules is T shape
XeF3+ :
The central atom : Xenon
The electronic configuration of Xenon is [Kr] 4d¹⁰ 5s² 5p⁶
The number of valence electrons in Xenon is 8
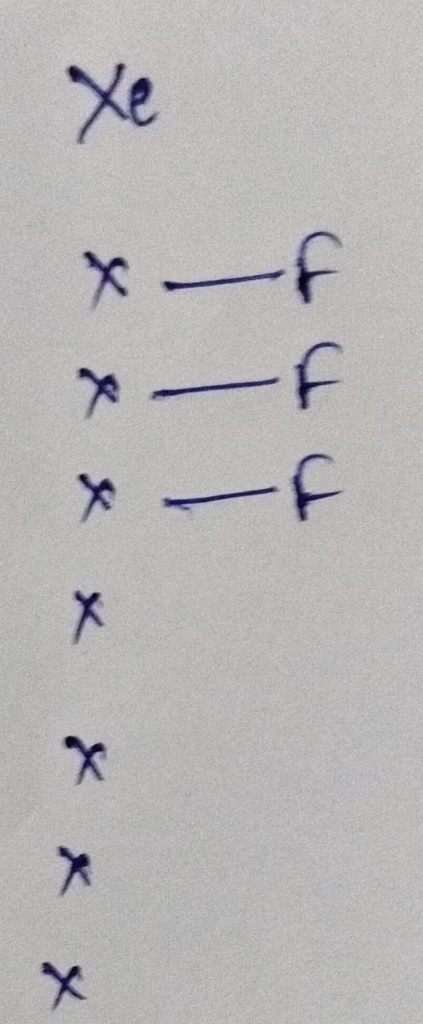
Hybridization : Number of bonds + lone pairs = 3 bonds +2 lone pairs = 5 { Sp3d hybridization }
The shape of XeF3+ molecule is T shape
Practice question :
Which of the following pairs of species have identical shapes ?
(a) NO2 and NO2–
(b) PCl5 and BrF5
(c) XeF4 and ICI4–
(d) TeCI4 and XeO4
Answer :
Correct answer : option (C)
XeF4 :
The central atom : Xenon
The electronic configuration of Xenon is [Kr] 4d¹⁰ 5s² 5p⁶
The number of valence electrons in Xenon is 8
- The Hybridization of XeF4
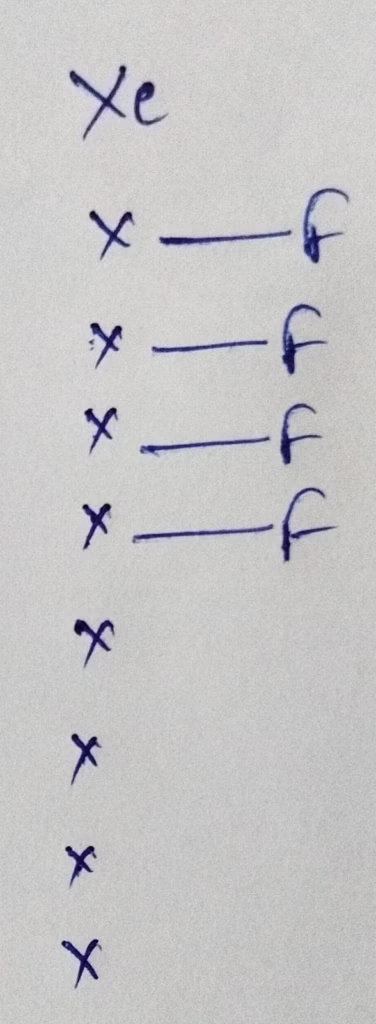
Hybridization : Number of bonds + lone pairs = 4 bonds + 2 lone pairs = 6 [ sp3d2 hybridization]
The shape of XeF4 molecule is T shape
ICl4– :
The central atom : Iodine
The electronic configuration of Iodine is [Kr] 4d¹⁰ 5s² 5p⁵
The number of valence electrons in iodine is 7
- The hybridization of ICl4–
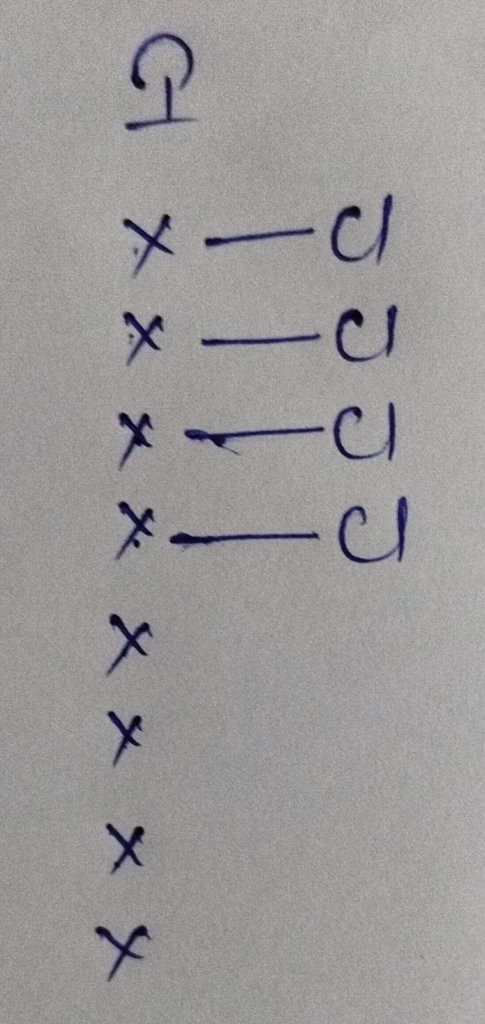
Hybridization : Number of bonds + lone pairs = 4 bonds+ 2lone pairs = 6 [sp3d2 hybridization ]
The shape of ICl4– molecule is T shape
2. Which is not correctly matched ?
(a) XeO3 – Trigonal bipyramidal
(b) CIF3 – bent T – shape
(c) XeOF4 – Square pyramidal
(d) XeF2 – Linear shape
Answer :
Correct answer : Option (a)
XeO3 :
The central atom is Xenon
The electronic configuration is [Kr] 4d¹⁰ 5s² 5p⁶
The number of valence electrons in Xenon is 8
- The hybridization of XeO3 molecule
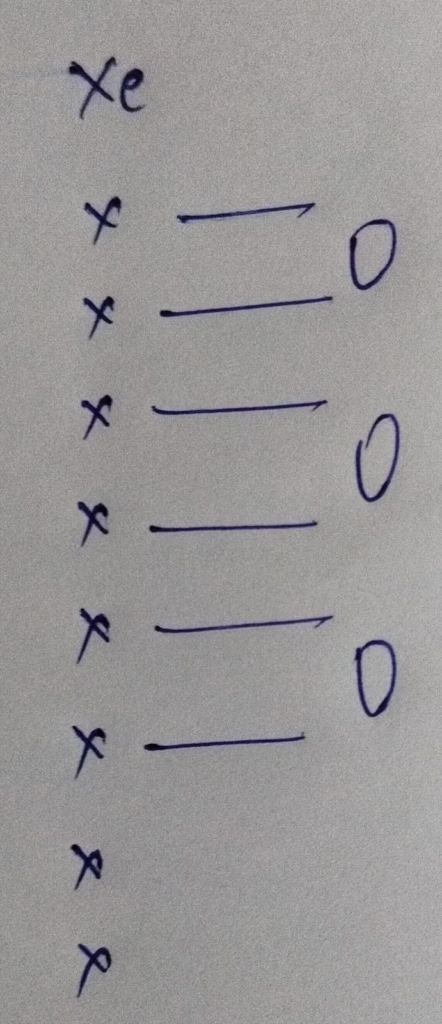
Hybridization : Number of bonds + Lone pair = 3 bonds + 1 lone pair = 4 [ sp3 hybridization ]
The shape XeO3 molecule is Trigonal pyramidal not the trigonal bipyramidal
3. In which of the following pairs, both the species have the same hybridization ?
(I) SF4 , XeF4
(II) I3– , XeF2
(III) ICl4+, SiCl4
(IV) ClO3–, PO43-
(a) I, II
(b) II, III
(c) II, IV
(d) I, II, III
Answer :
I3– :
The central atom : Iodine
The electronic configuration of iodine is [Kr] 4d¹⁰ 5s² 5p⁵
The number of valence electrons in iodine is 7
- The hybridization of I3–
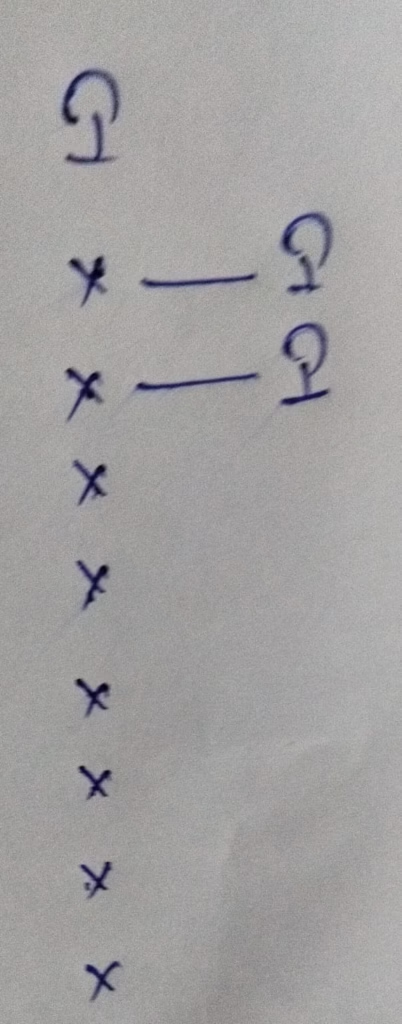
Hybridization : Number of bonds + Lone pair = 2 bonds + 3 lone pair = 5 [ sp3d hybridization ]
XeF2 :
The central atom is Xenon
The electronic configuration is xenon is [Kr] 4d¹⁰ 5s² 5p⁶
The number of valence electrons in Xenon is 8
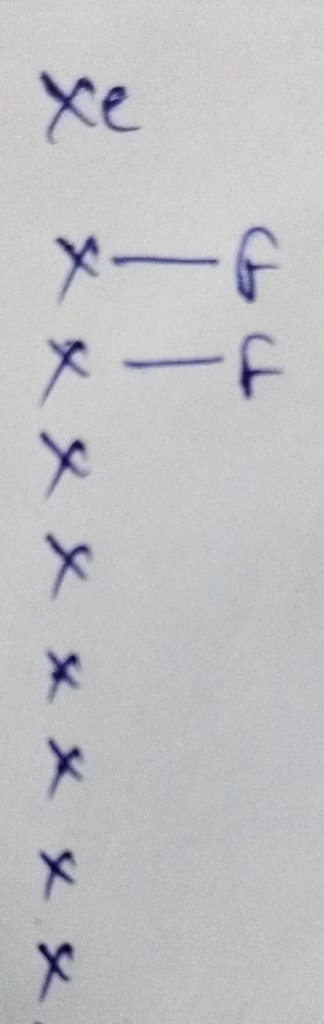
Hybridization : Number of bonds + Lone pair = 2 bonds + 3 lone pair = 5 [ sp3d hybridization ]
CIO3– :
The central atom : Chlorine
The electronic configuration of chlorine [Ne] 3s² 3p⁵
The number of valence electrons in chlorine is 7
- The hybridization of CIO3– molecule
Hybridization : Number of bonds + Lone pair = 3 bonds + 1 lone pair = 4 [sp3 hybridization ]
PO43- :
The central atom : Phosphorus
The electronic configuration of phosphorus : [Ne] 3s² 3p³
The number of valence electrons in phosphorus is 5
- The hybridization of PO43- molecule
Hybridization : Number of bonds + Lone pair = 4 bonds + 0 lone pair = 4 [ sp3 hybridization ]
Correct Option : (c)
Visit more PYQs on http://jeestudyhub.in