Question :
The set which does not have ambidentate ligand(s) is
Options :
(a) C2O42-, ethylene diamine, H2O
(b) EDTA4-, NCS–, C2O42-
(c) NO2–, C2O42-, EDTA4-
(d) C2O42-, NO2–, NCS–
JEE Main 2023 Co – ordination Compounds with Solution
Answer :
Correct answer : Option (a)
Ambidentate ligand :
- The ligand which have donor atoms but in forming complexes only one donor atom is attached to the metal atom at a given time. Such ligands are called ambidentate ligands.
Examples :
- M NO2 [ Nitrito – N ] and M ONO [ Nitrito – O ]
- M CN [ Cyano – C ] and M NC [ Isocyano ]
In above question
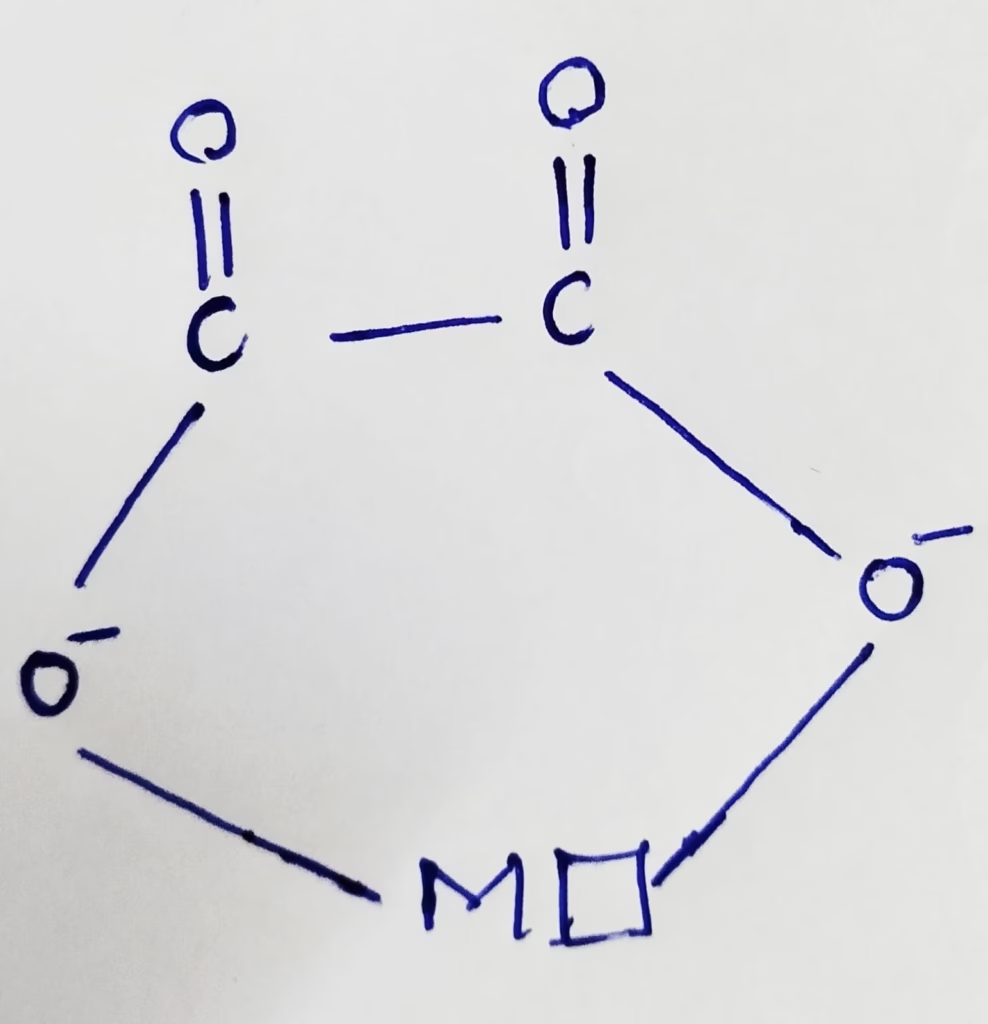
C2O42- :
- C2O42- is bidentate which have two donor atoms and have the ability to link with central metal at two positions
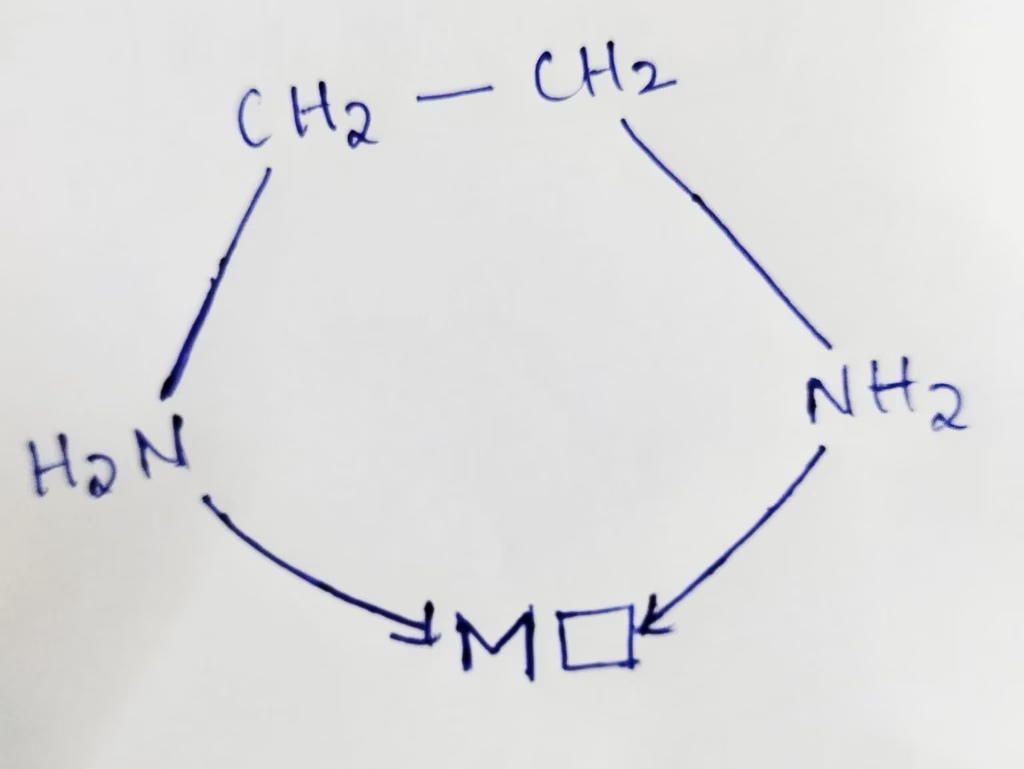
en ( ethylene diamine ) :
- en ( ethylene diamine ) is bidentate which have two donor atoms of Nitrogen and have the ability to link with central metal atom
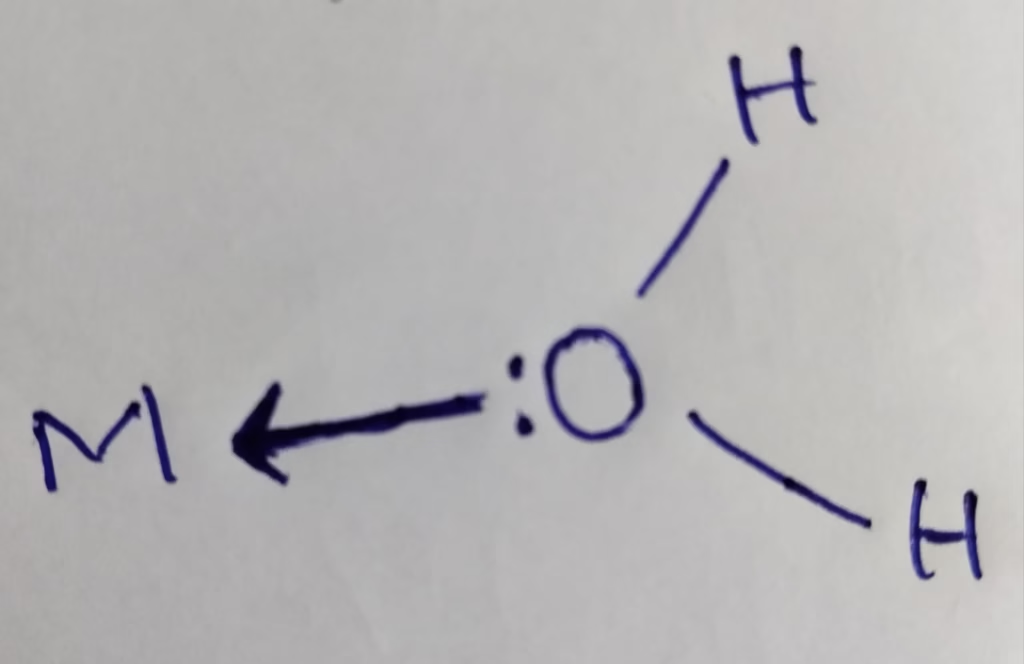
H2O :
- H2O is Mono dentate which have one donor atom that donates only one electron pair to central metal atom
hence , in Option (a) No ambidentate ligand is present
Concept Involved :
- Ambidentate Ligands (Coordination Chemistry)
Examples for Practice :
Find the central atom of ( i ) oxidation number ( ii) Co ordination number (iii) Effective atomic number
- K4[ Fe(CN)6 ]
i) Oxidation state of Central metal atom :
4 [ +1] + x + 6( – 1) = 0
x – 2 = 0
x = 2
ii) Co ordination number of central metal atom :
Co – ordination number : Number of ligands x denticity = 6×1 = 6
iii) Effective atomic number [ EAN ] :
= Atomic number of Central metal atom – oxidation number + 2 x co ordination number
= 26 – 2 + 2 x 6
= 24 + 12 = 36 [ stable ]
2 .K3 [ CO ( C2O4)3 ]
i) Oxidation state of central metal atom
3 [ +1] + x + 3( -2) = 0
3 + x – 6 = 0
x = +3
ii) Co – ordination number of central metal atom :
Co – ordination number : Number of ligands x denticity = 3 x 2
iii) Effective atomic number [ EAN ] of K3 [ CO ( C2O4)3 ] :
= Atomic number of Central metal atom – oxidation number + 2 x co ordination number
= 27 – 3 + 2 x 6 = 36 [ stable ]
3. [ Fe ( CN6]-3
i) Oxidation state of Central metal atom :
x + 6 ( – 1) = – 3
x – 6 = -3
x = +3
ii) Co – ordination number of central metal atom :
Co – ordination number : Number of ligands x denticity = 6 x 1
iii) Effective atomic number [ EAN ] of [ Fe ( CN6]-3
= Atomic number of Central metal atom – oxidation number + 2 x co ordination number
= 26 – 3 + 2 x 6
= 23 + 12 = 35 [ stable ]
4. [ CO (NH3)6]Cl3
i) Oxidation state of Central metal atom :
= x + 6(0) + 3(-1) = 0
x = +3
ii) Co – ordination number of central metal atom :
Co – ordination number : Number of ligands x denticity = 6 x 1 = 6
iii) Effective atomic number [ EAN ] of [ CO (NH3)6]Cl3
= Atomic number of Central metal atom – oxidation number + 2 x co ordination number
27 – 3 + 2 x 6 = 24 + 12 = 36
5. The oxidation number of Co in [ Co(en)3]2(SO4)3 is
Options :
(a) +2
(b) +4
(c) +3
(d) +6
Answer :
oxidation number of Co in [ Co(en)3]2(SO4)3
2x + 3(0) + 3( -2) = 0
2x = 6
x = +3
Correct Answer : Option (c)
6. The oxidation state of Cr in [ Cr(NH3)4Cl2]+
Options :
(a) +1
(b) 0
(c) +3
(d) +2
Answer :
The oxidation state of Cr in [ Cr(NH3)4Cl2]+
x + 4(0) + 2(-1) = +1
x = +3
Visit more PYQs on https://jeestudyhub.in/