This JEE Main 2023 Chemical Bonding PYQ with Solution helps students learn hybridization and VSEPR Theory for JEE Main exam. Practicing PYQs improves problem- solving skills and helps students prepare better for the JEE Main.
Question :
Match List – I with List – II
| List – I (Molecule/ions) | List – II (No. of lone pairs on central atom) |
|---|---|
| (A) IF7 | I) Three |
| (B) ICl–4 | II) One |
| (C) XeF6 | III) Two |
| (D) XeF2 | Iv) Zero |
Options :
(a) A – II, B – III, C – IV, D – I
(b) A – IV, B – III, C – II, D – I
(c) A – II, B – I , C – IV, D – III
(d) A – IV, B – I, C – II, D – III
JEE Main 2023 Chemical Bonding PYQ with Solution
Answer :
IF7 :
The central atom is Iodine
The electronic configuration of iodine is [Kr]4d105s25p5
The Number of valence electrons present in iodine is 7
- Hybridization of IF7 molecule
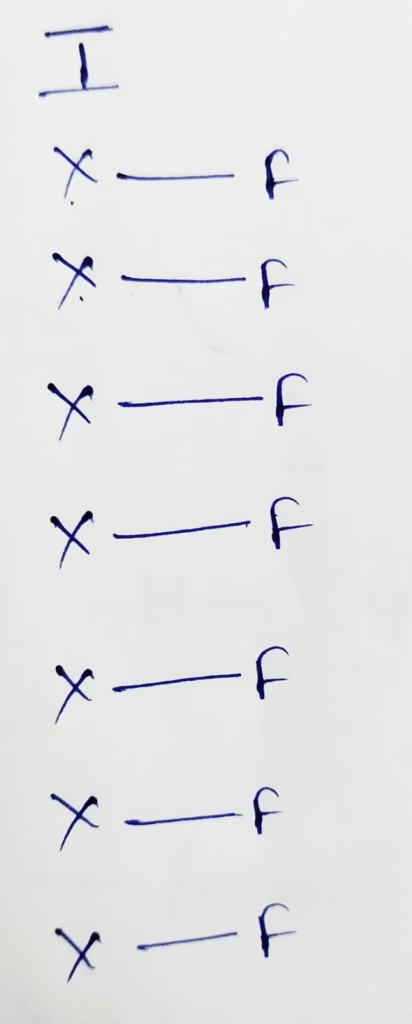
Hybridization : Number of bonds + Lone pair = 7 + 0 Lone pair
The Number of Lone pair present in IF7 molecule is ‘zero’
ICl–4 :
The central atom is iodine
The electronic configuration is [Kr]4d105s25p5
The valence electrons present in iodine is 7
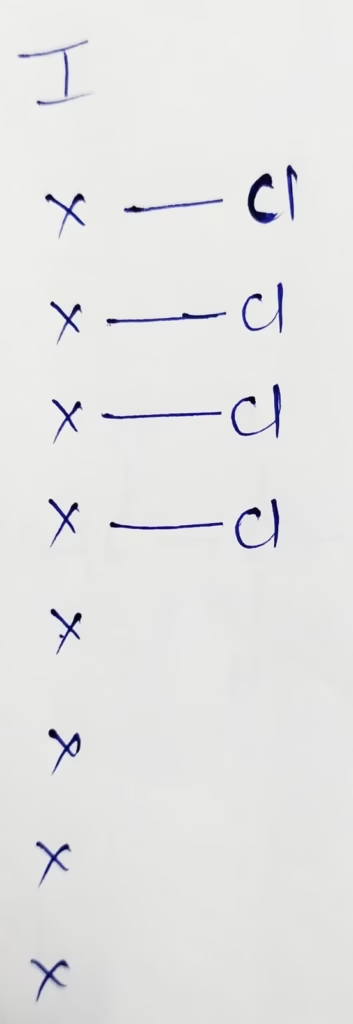
Hybridization : Number of + Lone pair = 4 + 2 lone pair = 6 [sp3d2]
The Number of lone pair present in ICl4– molecule is ‘Two’
XeF6 :
The central atom is Xenon
The electronic configuration of Xeon is [Kr]4d105s25p6
The number of valence electrons present in Xenon is 8
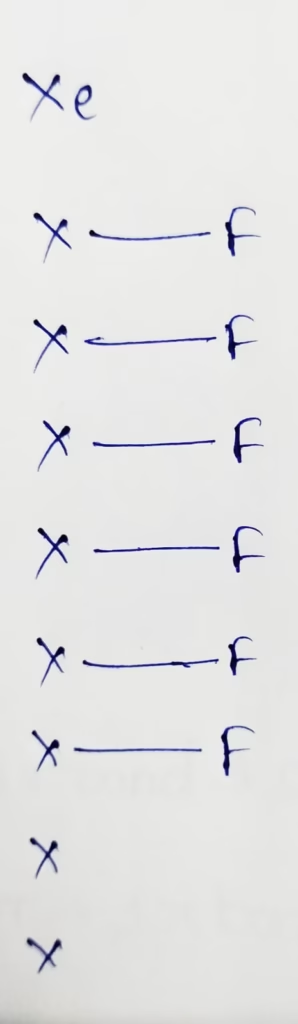
Hybridization : Number of bonds + Lone pair = 6 + 1lone pair
The number of lone pair present in XeF6 molecule is ‘one’
XeF2 :
The central atom is Xenon
The electronic configuration of Xenon is [Kr]4d105s25p6
The number of valence electrons present in Xenon is 8
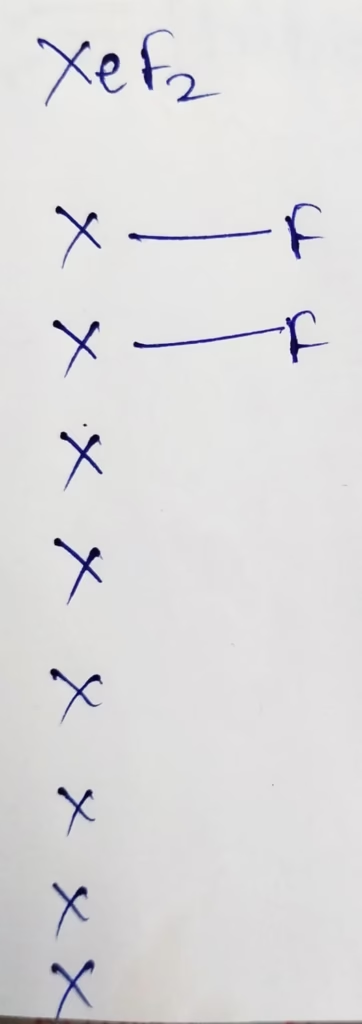
Hybridization : Number of bonds + Lone pair = 2 + 3 lone pair
The number of lone pair present in XeF2 molecule is ‘Three’
Correct answer : Option(b)
Concept Involved :
- Hybridization
- VSEPR Theory
Visit more PYQ on http://jeestudyhub.in
Practice Questions :
- The hybridization of central iodine atom in IF5, I3– and I3+ respectively :
(a) sp3d2, sp3d , sp3
(b) sp3d , sp3d, sp3
(c) sp3d2, sp3d2, sp3
(d) sp3d, sp3d2, sp3
Answer :
IF5 :
The central atom : Iodine
Electronic configuration : [Kr] 4d¹⁰ 5s² 5p⁵
The number of valence electrons in Iodine is 7
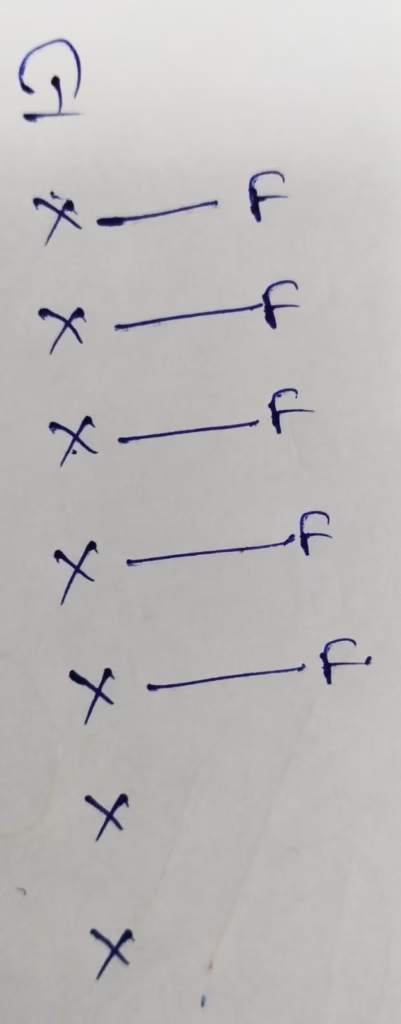
Hybridization : Number of bonds + Lone pair = 5 + 1 lone pair = 6 [ sp3d2 hybridization ]
I–3 :
The central atom : Iodine
Electronic configuration : [Kr] 4d¹⁰ 5s² 5p⁵
The number of valence electrons in iodine is 7
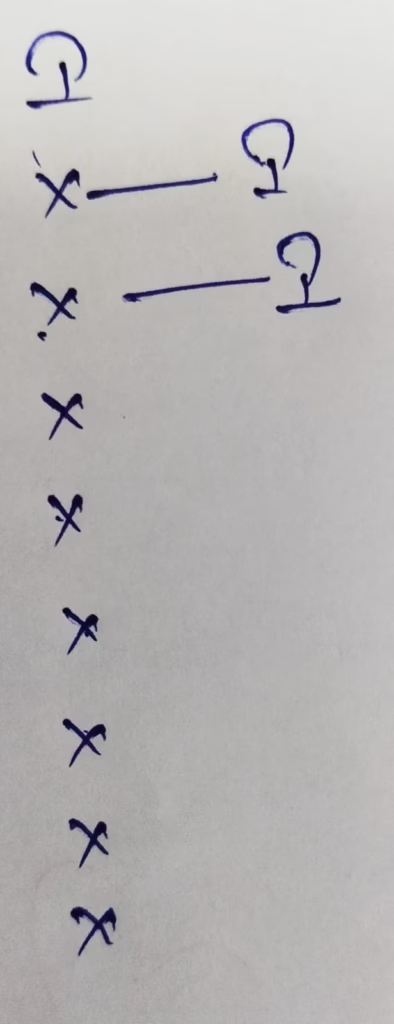
Hybridization : Number of bonds + Lone pair = 2 + 3 lone pair = 5 [ sp3d hybridization ]
I3+ :
The central atom : Iodine
The electronic configuration : [Kr] 4d¹⁰ 5s² 5p⁵
The number of valence electrons in iodine is 7
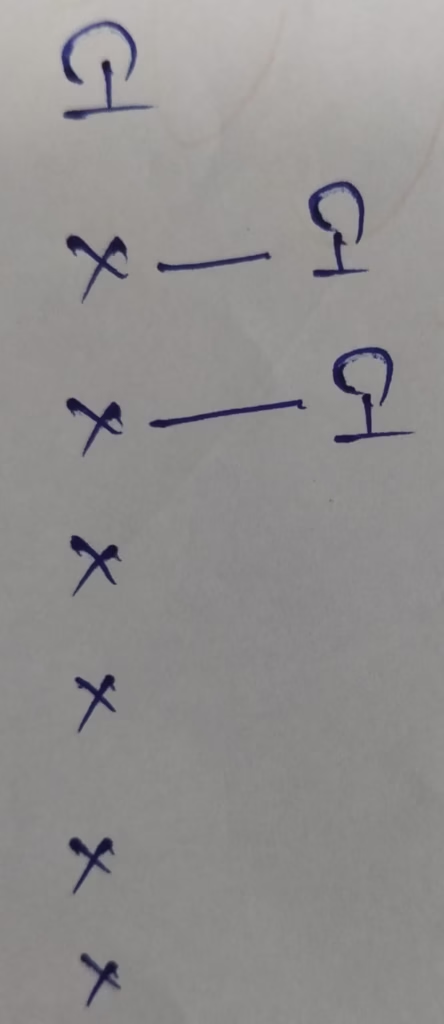
Hybridization : Number of bonds + Lone pair = 2 + 2 lone pair = 4 [sp3 hybridization ]
Correct answer : Option (a)
Concept involved :
- Hybridization
2. Which bonds are formed by a carbon atom with sp2 hybridization ?
(a) 4 bonds
(b) 2 bonds and 2 bonds
(c) 1 bonds and 3 bonds
(d) 4 bonds
Answer :
1 bond and 3 bonds = sp2 hybridization
Correct answer : option (c)
3. The H——O——H bond angles in H3O+ are approximately 107o The orbitals used by oxygen in these bonds are best described as :
(a) p orbitals
(b) sp hybrid orbitals
(c) sp2 hybrid orbital
(d) sp3 hybrid orbital
Answer :
The central atom is Oxygen
The electronic configuration : [He] 2s² 2p⁴
The number of valence electrons present in oxygen is 6
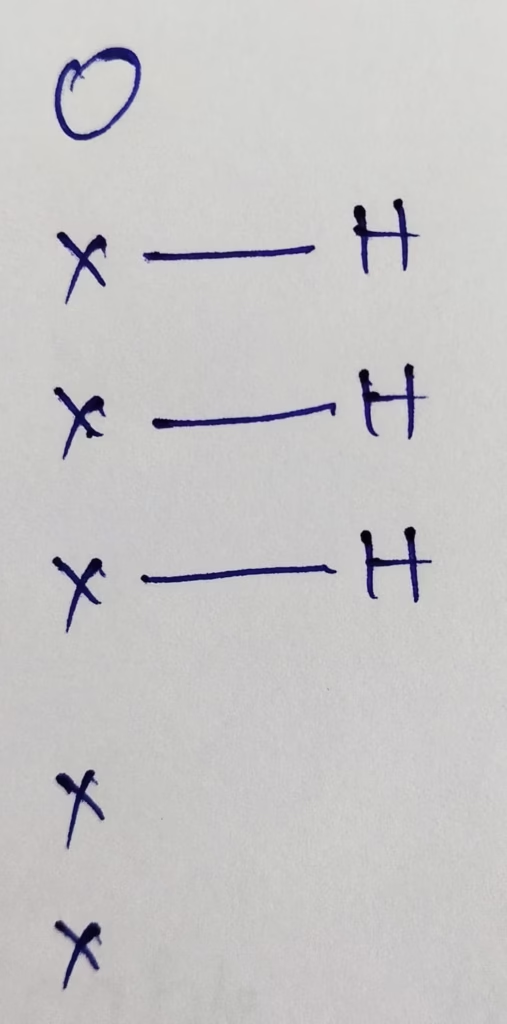
Hybridization : Number of bonds + Lone pair = 3 bonds + 1 lone pair = 4 [ sp3 hybridization ]