This Chemical Bonding JEE Main 2018 PYQ with solution helps student understand Hybridization and Molecular Geometry. Solving More PYQs helps student improve problem-solving skills.
Question :
Identify the pair in which the geometry of the species is T – shape and square pyramidal respectively
Options :
(a) ICI2– and ICI5
(b) IO3 and IO2F2–
(c) ClF3 and IO4–
(d) XeOF2 and XeOF4
Chemical Bonding JEE Main 2018 PYQ with solution
Answer :
Correct answer : Option (d)
XeOF2 :
The central atom is Xenon
The electronic configuration of Xenon is [Kr] 4d¹⁰ 5s² 5p⁶
The number of valence electrons in Xenon is 8
- The hybridization of Xenon in XeOF2
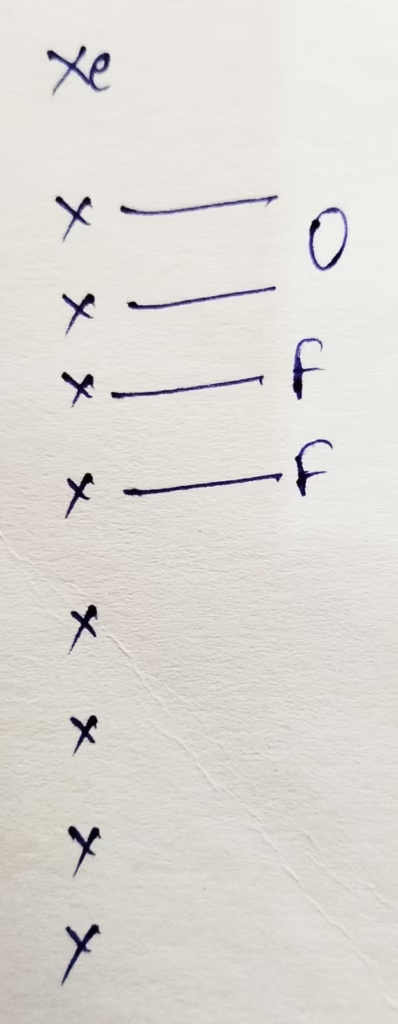
Hybridization : Number of bonds + Lone pair = 3 bonds + 2 lone pair = 5 [ sp3d hybridization ]
The shape of XeOF2 molecule is T – shape
XeOF4 :
The central atom : Xenon
The electronic configuration of Xenon is [Kr] 4d¹⁰ 5s² 5p⁶
The number of valence electrons in Xenon is 8
- The hybridization of Xenon in XeOF4
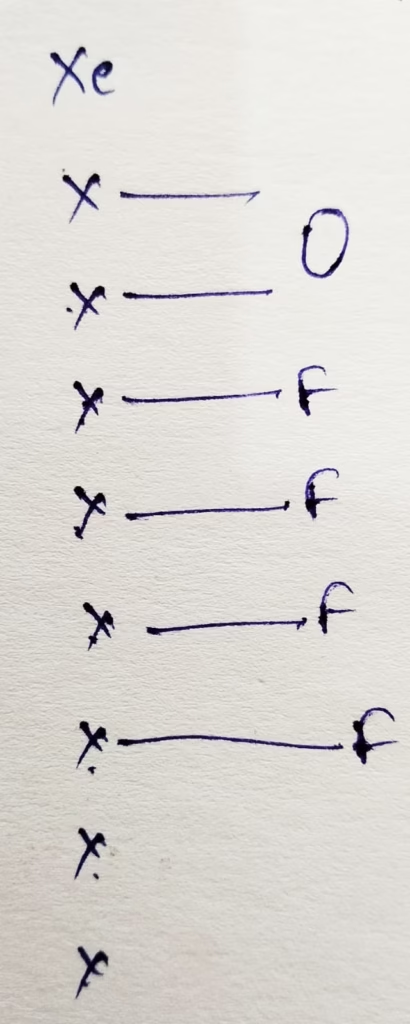
Hybridization : Number of bonds + Lone pair = 5 bonds + 1 lone pair = 6 [ sp3d2 hybridization ]
The shape of XeOF4 molecule is square pyramidal.
Concept Involved :
- Hybridization
Practice Questions :
- The pair of species with similar shape :
(a) PCl3, NH3
(b) CF4, SF4
(c) PbCl2, CO2
(d) PF5, IF5
Answer :
Correct answer : Option (a)
The central atom : Phosphorus
The electronic configuration of phosphorus is [Ne] 3s² 3p³
The number of valence electrons in phosphorus is 5
- The hybridization of phosphorus in PCl5 :
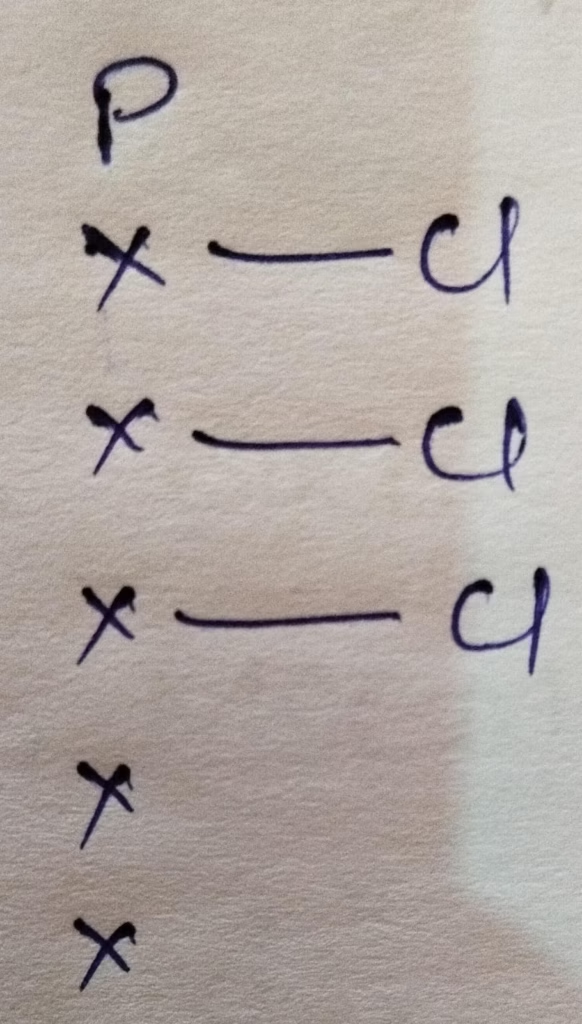
Hybridization : Number of bonds + Lone pair = 3 bonds + 1 lone pair = 4 [sp3 hybridization ]
The shape of PCl3 molecule is Trigonal pyramidal
NH3 :
The central atom : Nitrogen
The electronic configuration of nitrogen is [Ne] 3s² 3p³
The number of valence electrons in nitrogen is 5
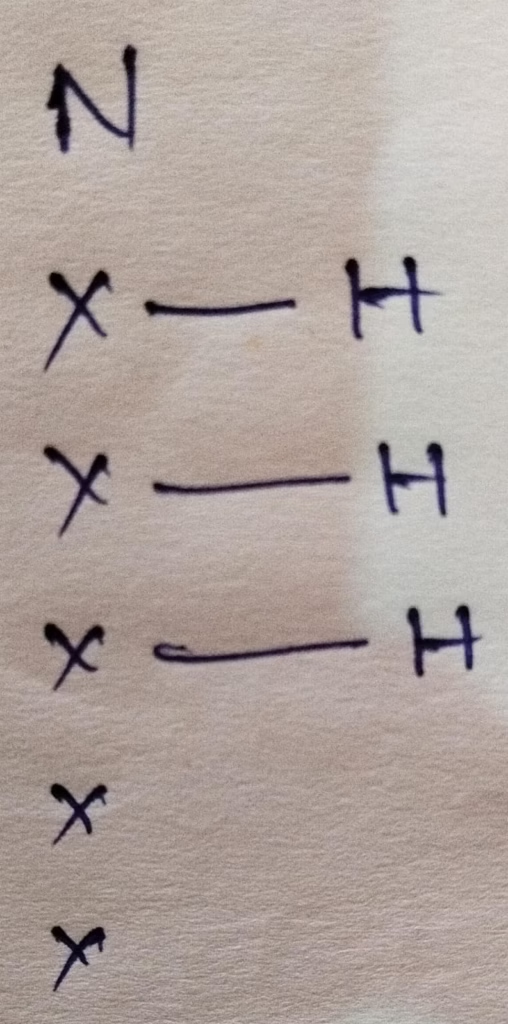
Hybridization : Number of bonds + Lone pair = 3 bonds + 1 lone pair = 4 [sp3 hybridization ]
The shape of NH3 molecule is Trigonal pyramidal
The PCl3, NH3 pair of species has same shape
2. The BCl3 is a planar molecule where as NCl3 is pyramidal because :
(a) B-Cl bond is more polar than N-Cl bond
(b) N – Cl bond is more covalent that B – Cl bond
(c) nitrogen atom is smaller than boron atom
(d) BCl3 has no lone pair of electrons but NCl3 has a lone pair of electrons
Answer :
Correct answer : Option (d)
BCl3 :
The central atom : Boron
The electronic configuration of boron is [He] 2s² 2p¹
The number of valence electrons present in boron is 3
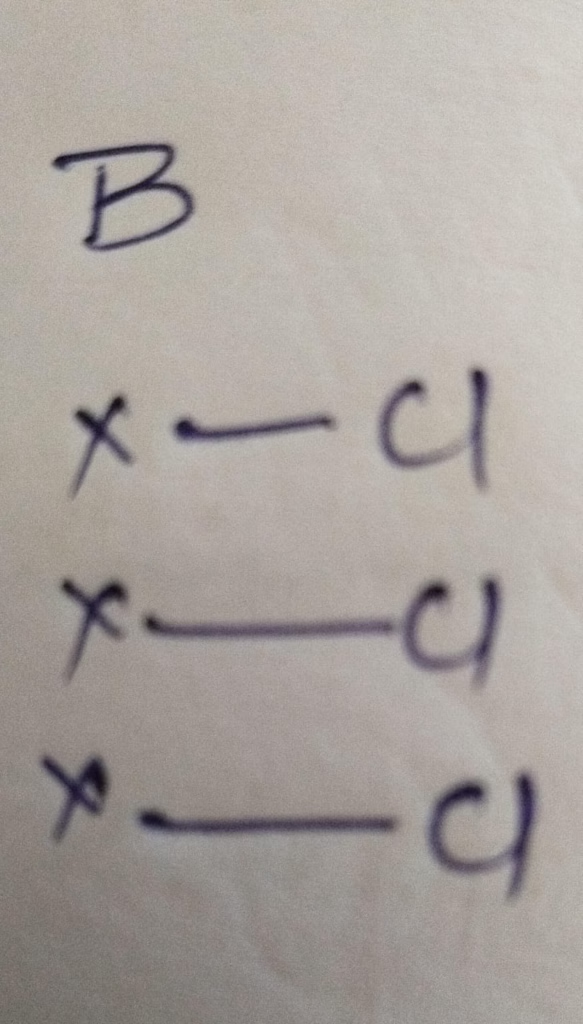
Hybridization : Number of bonds + lone pair = 3 bonds + 0 lone pair = 3 [ sp2 hybridization ]
The Number of lone pair present in BCl3 molecule is 0 lone pair
NCl3 :
The central atom : Nitrogen
The electronic configuration of nitrogen is [Ne] 3s² 3p³
The number of valence electrons present in nitrogen is 5
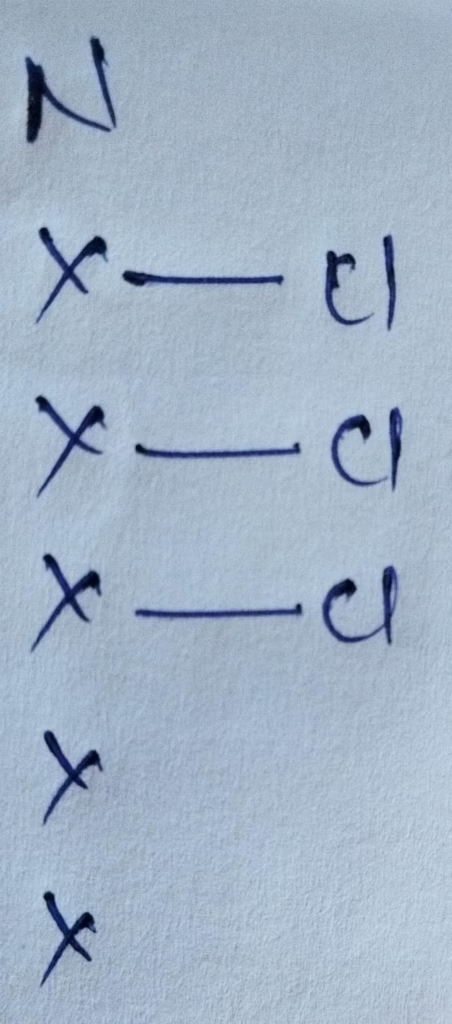
Hybridization : Number of bonds + Lone pair = 3 bonds + 1 lone pair = 4 [ sp3 hybridization ]
The number of lone pair present in NCl3 molecule is 1 lone pair
3. Correct statement regarding molecules SF4, CF4 and XeF4 are :
Options :
(a) 2, 0, and 1 lone pairs of central atom respectively
(b) 1, 0 and 1 lone pair of central atom respectively
(c) 0, 0 and 2 lone pair of central atom respectively
(d) 1, 0 and 2 lone pair of central atom respectively
Answer :
SF4 :
The central atom is Sulphur
The electronic configuration of Sulphur is [Ne]3s23p4
The Number of valence electrons present in Sulphur is 6
- Hybridization of Sulphur in SF4
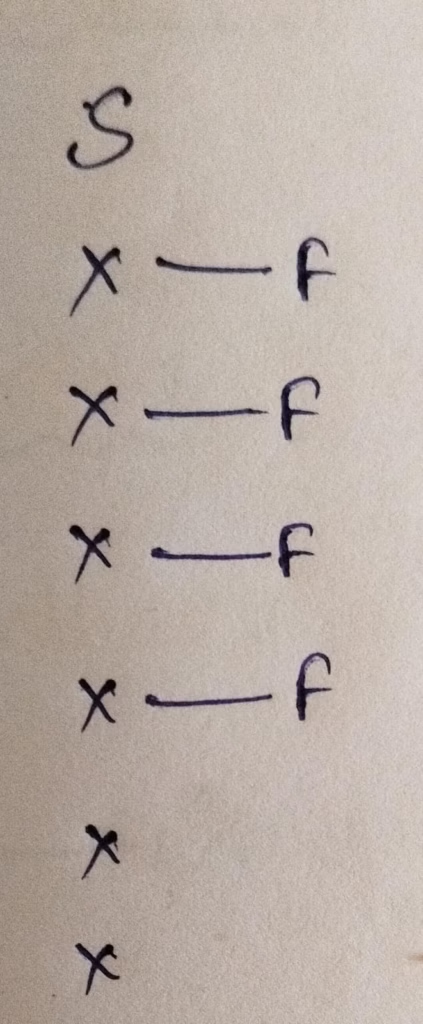
Hybridization : Number of bonds + Lone pair = 4 bonds + 1 lone pair = 5 [ sp3d hybridization ]
The number of lone pair present in central atom of SF4 molecule is 1 lone pair
CF4 :
The central atom is Carbon
The electronic configuration of Carbon is [He] 2s² 2p²
The Number of valence electrons present in Carbon is 4
- Hybridization of carbon in CF4 :
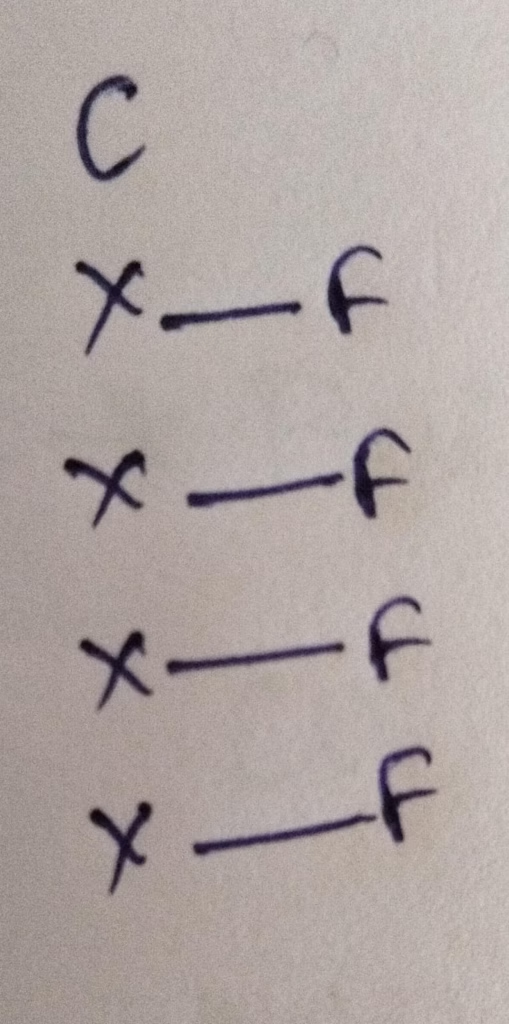
Hybridization : Number of bonds + Lone pair = 4 bonds + 0 lone pair = sp3 hybridization
The number of lone pair present in central atom of CF4 is 0 lone pair
XeF4 :
The central atom is Xenon
The electronic configuration of Xenon is [Kr] 4d¹⁰ 5s² 5p⁶
The Number of valence electrons present in Carbon is 8
- Hybridization of Xenon in XeF4
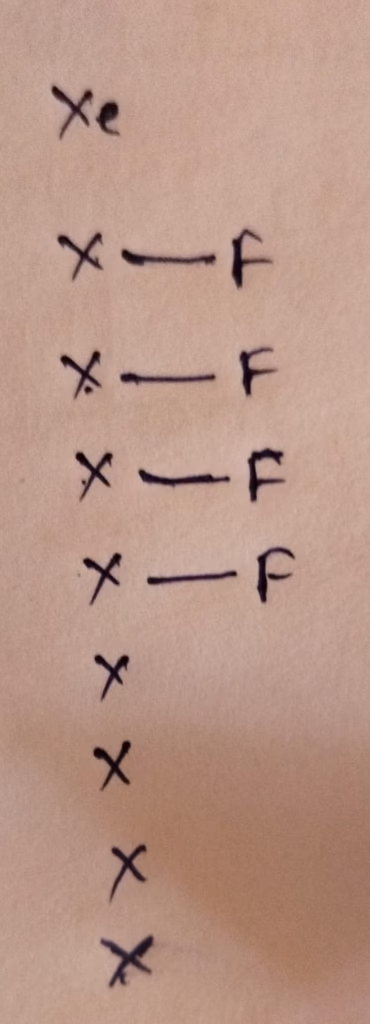
Hybridization : Number of bonds + Lone pair = 4 bonds + 2 lone pair = 6 [ sp3d2 hybridization]
The number of lone pair present in central atom of XeF4 is 2
Visit more PYQs on http://jeestudyhub.in