Chemical Bonding JEE Main 2023 PYQ with Solution is explained here with a clear explanation. This question helps students understand Hybridization and VSEPR Theory for the JEE Main exam.
Question :
Match List I with List II
| List I | List II |
|---|---|
| A) XeF4 | I) See-saw |
| B) SF4 | II) Square planar |
| C) NH+4 | III) Bent T- shaped |
| D) BrF3 | IV) Tetrahedral |
Choose the correct answer from the options given below :
(a) A – IV, B – III, C – II, D – I
(b) A – II, B – I, C – III, D – IV
(C) A – IV, B – I , C – II, D – III
(d) A – II, B – I, C – IV, D – III
Chemical Bonding JEE Main 2023 PYQ with Solution
Answer :
XeF4 :
The Central atom is Xeon
The electronic configuration of Xeon is [Kr]4d105s25p6
The Number of valence electrons present in Xeon is 8
- The Hybridization of Xeon in XeF4

Hybridization : number of bonds + lone pairs = 4 + 2 Lone pairs = 6[sp3d2 hybridization ]
The shape of XeF4 molecule is square planar
SF4 :
The Central atom is Sulphur
The electronic configuration of Sulphur is [Ne]3s23p4
The Number valence electrons present in Sulphur is 6
- The Hybridization of Sulphur in SF4

Hybridization : Number of bonds + lone pairs = 4 + 1 lone pair = 5[sp3d hybridization ]
The shape of SF4 Molecule is see-saw
NH+4 :
The central atom is Nitrogen
The electronic configuration of Nitrogen is 1s22s22p3
The valence electrons present in nitrogen is 5
- The hybridization of nitrogen in NH4+

Hybridization : Number of bonds + Lone pair = 4 + 0 lone pair = 4 [sp3 hybridization]
The shape of NH+4 molecule is tetrahedral
BrF3 :
The central atom is Bromine
The electronic configuration of Bromine is [Ar]3d104s24p5
The Number of valence electrons present in Bromine is 7
- The Hybridization of Bromine in BrF3
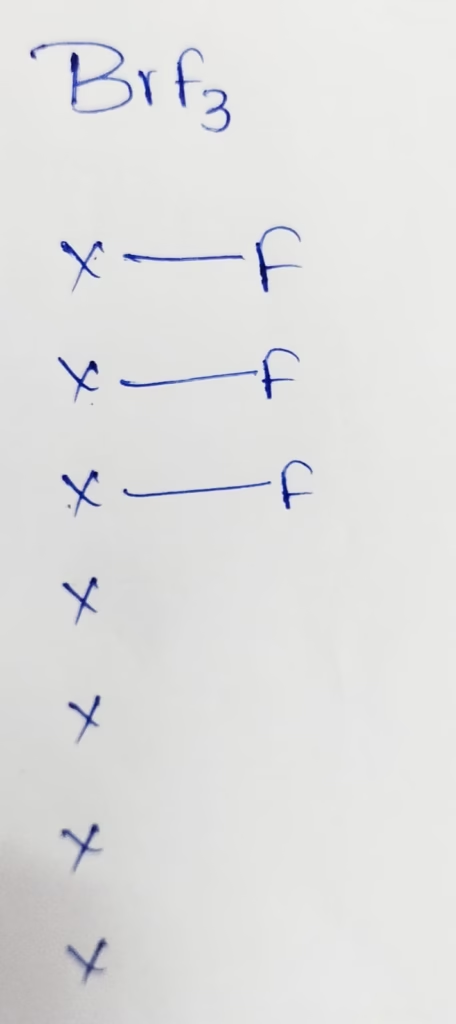
Hybridization : Number of bonds + Lone pair = 3 + 2lone pair = 5[sp3d hybridization ]
The shape of BrF3 molecule is T- shape
Correct Answer : Option (d)
Concept Involved :
- VSEPR Theory
- Hybridization
Visit more PYQS on http://jeestudyhub.in
Practice Questions :
1. The hybridization of the central atom in ICl2+ is :
Options :
(a) dsp2
(b) sp
(c) sp2
(d) sp3
Answer :
The central atom is iodine
The electronic configuration : [Kr]4d105s25p5
The number of valence electrons in iodine is 7
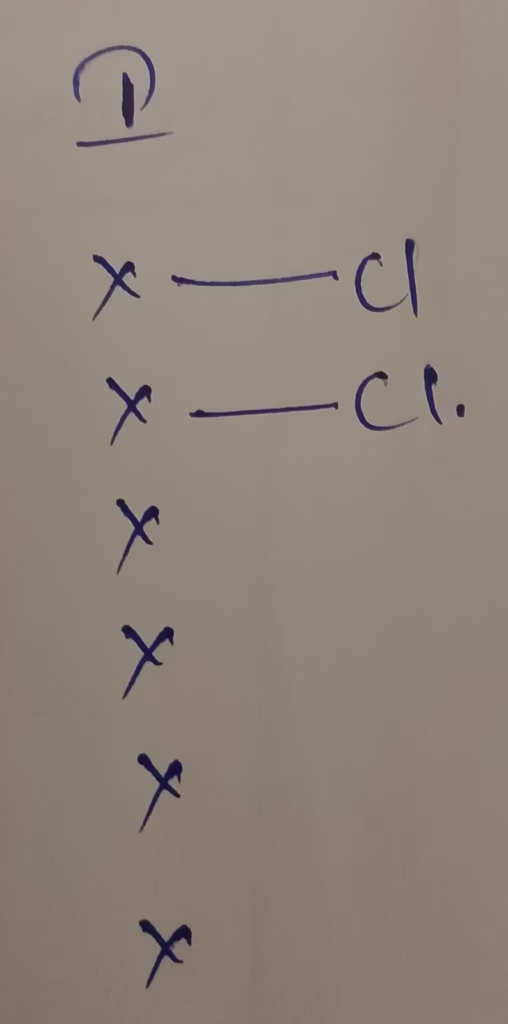
Hybridization : Number of bonds + Lone pair = 2 + 2 lone pair = 4 [sp3 hybridization ]
Correct answer : option (d)
Concept involved :
- Hybridization
2. What is the state of hybridization of Xe in cationic part of solid XeF6 ?
Options :
(a) sp3d3
(b) sp3d2
(c) sp3d
(d) sp3
Answer :
The central atom : Xenon
The electronic configuration : [Kr]4d105s25p6
The number of valence electrons in Xenon is 8
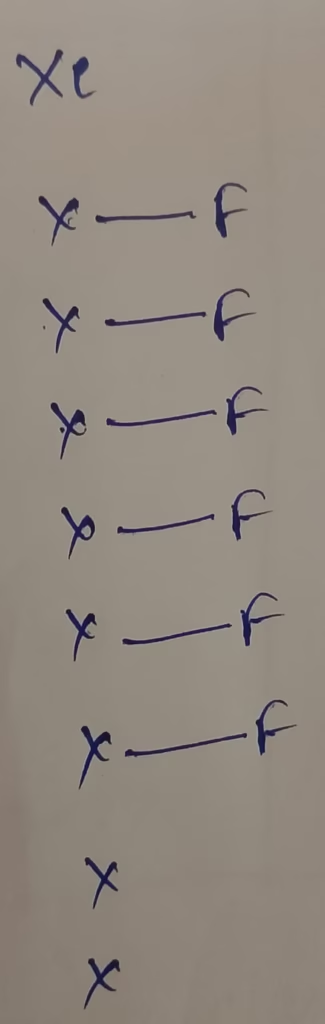
Hybridization : Number of bonds + Lone pair = 6 + 1 lone pair = 7 [sp3d2 hybridization]
Correct answer : Option (b)
Concept involved :
- Hybridization
3. Which one of the following is the correct set with respect to molecule, hybridization and shape ?
(a) BeCl2, sp2, linear
(b) BeCl2 , sp2, triangular planar
(c) BCl3, sp2, triangular planar
(d) BCl3, sp3, tetrahedral
Answer :
Correct answer : Option (c)
The central atom is Boron
The electronic configuration of boron is [He]2s22p1
The number of valence electrons in boron is 3
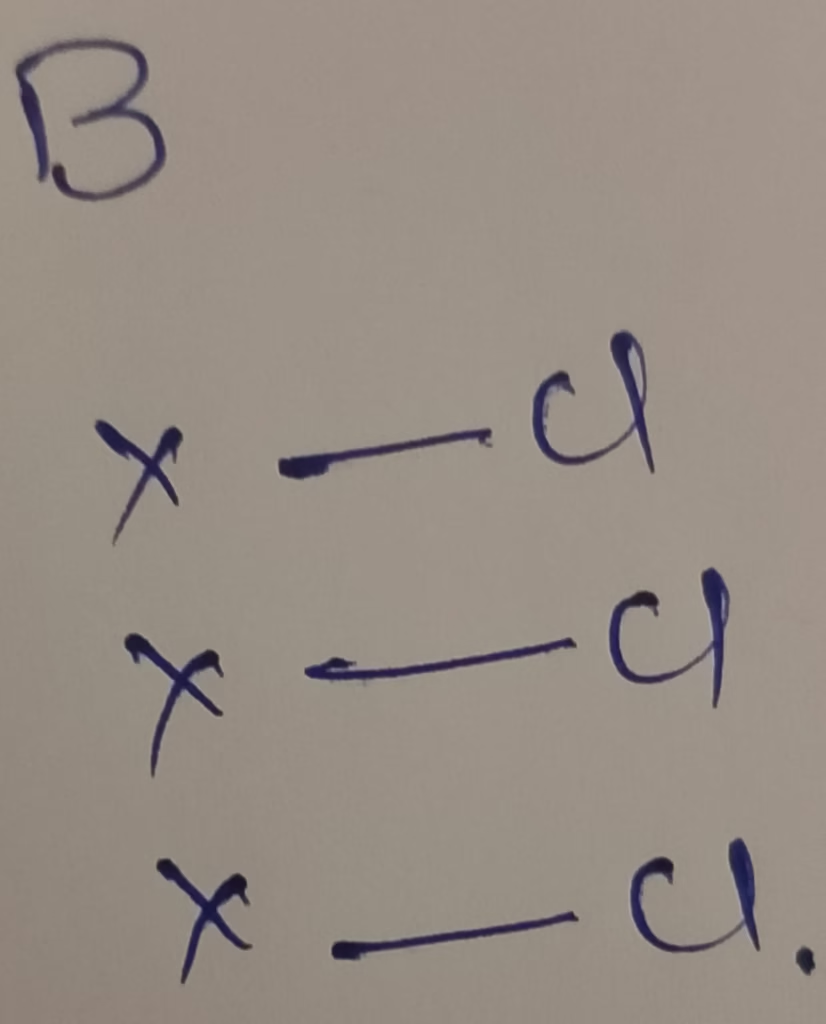
Hybridization : Number of bonds + Lone pair = 3 + 0 lone pair = 3 bonds [sp2 hybridization ]
The shape of BCl3 molecule is triangular planar
concept involved :
- Hybridization
Conclusion :
- Practicing Chemical Bonding JEE Main 2023 PYQ with Solution help student understand bonding concepts. and improves their performance in exam.
