Question :
Number of lone pairs of electrons in the central atom of SCl2 , O3 , ClF2 and SF6 respectively are
Options :
(a) 0 ,1 ,2 and 2
(b) 2, 1 , 2 and 0
(c) 1, 2, 2 and 0
(d) None of these
Chemical Bonding JEE Main 2022 PYQ with Detailed Solution.
Answer :
SCl2 :
The central atom is Sulphur
The electronic configuration of Sulphur is [Ne]3s23p4
The Number of valence electrons present in Sulphur is 6
- Hybridization of Sulphur in SCl2
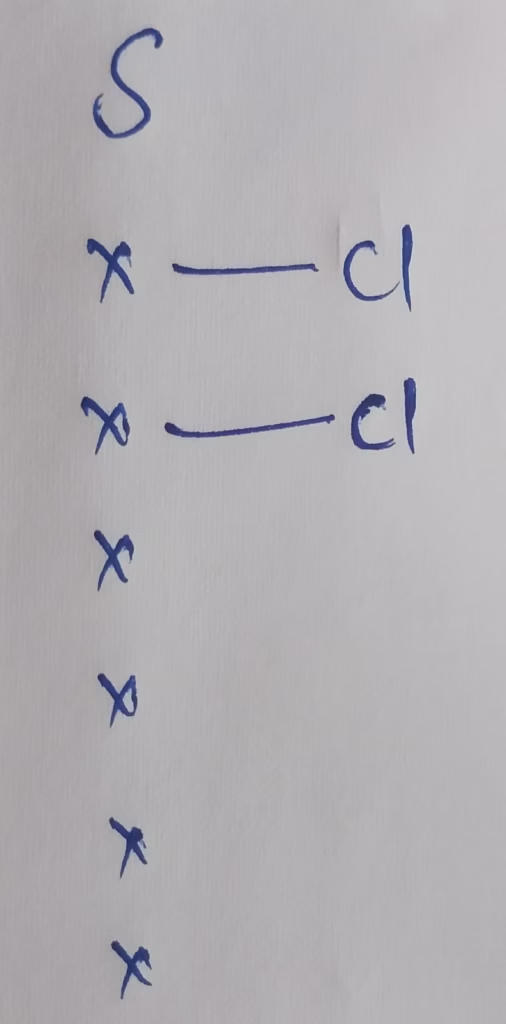
Hybridization : Number of bonds + Lone pairs = 2 + 2 lone pairs
The Number of Lone pairs present in SCl2 Molecule : 2
O3 :
The Central atom is Oxygen
The electronic Configuration of Oxygen is 1s22s22p4
The Number of valence electrons present in Oxygen is 6
- The Hybridization of Oxygen in O3
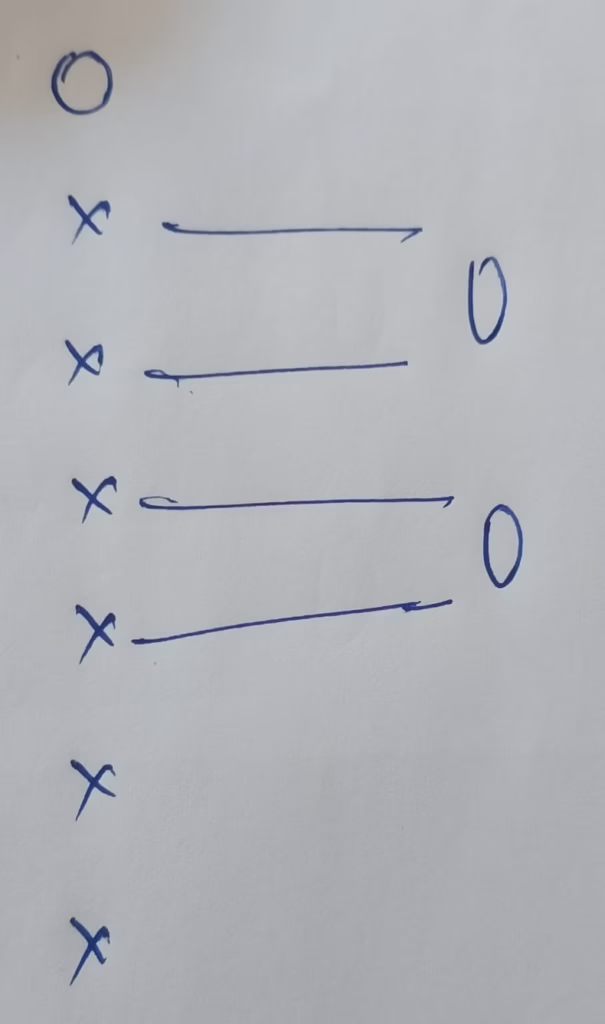
Hybridization : Number of bond + Lone pairs = 2 bonds + 1Lone pair
Number of Lone pairs present in oxygen : 1
ClF3 :
The central atom is Cl
The Electronic configuration of chlorine is [Ne]3s23p5
The Number of Valence electrons present in chlorine is 7
- The Hybridization of chlorine in ClF3

Hybridization : Number of bonds + Lone pairs = 3 bonds + 2 lone pair
Number of Lone pairs present in ClF3 Molecule is 2
SF6:
The central atom is Sulphur
The electronic configuration of Sulphur is [Ne]3s23p4
The Number valence electrons present in Sulphur is 6
- The Hybridization of Sulphur in SF6

Hybridization : Number of bonds + Lone pair = 6 bonds + 0 lone pair
Number of Lone pairs present in SF6 Molecule is 0
Correct Answer : Option (b)
Concept Involved :
- VSEPR Theory
- Hybridization
Visits more PYQs jeestudyhub.in
Practice Questions :
- The hybridization of atomic orbitals of N in NO2+, NO3–, and NH4+ are respectively :
(a) sp, sp2, sp3
(b) sp, sp3, sp2
(c) sp2, sp, sp3
(d) sp2, sp3, sp
Answer :
NO2+
The central atom is Nitrogen
The electronic configuration of Nitrogen is 1s22s22p3
The valence electrons present in nitrogen is 5
- The hybridization of nitrogen in NO2+
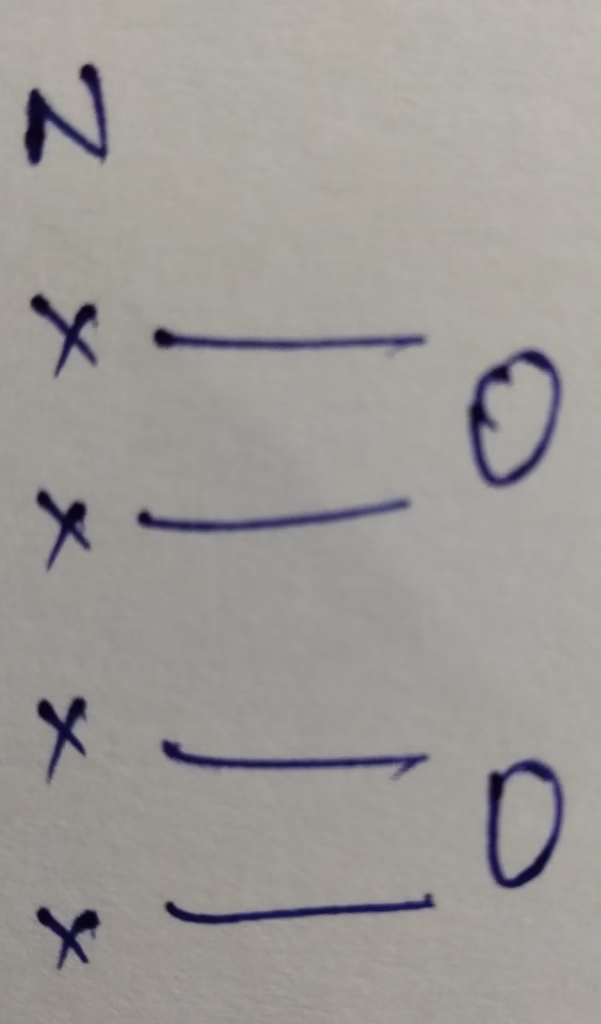
Hybridization : Number of bonds + Lone pair = 2 bonds + 0 Lone pair = 2 [ sp hybridization ]
NO3– :
The central atom is Nitrogen
The electronic configuration of Nitrogen is 1s22s22p3
The valence electrons present in nitrogen is 5
- The hybridization of nitrogen in NO3–
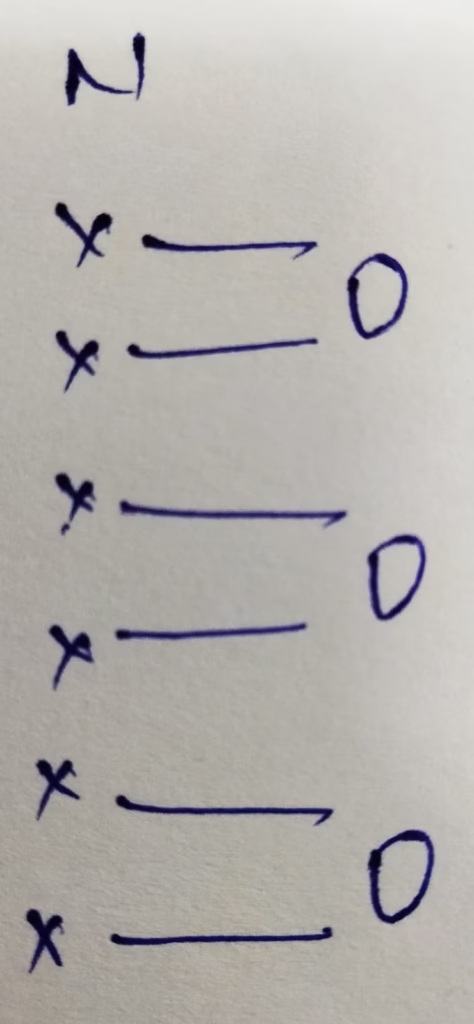
Hybridization : Number of bonds + Lone pair = 3 bonds + 0 lone pair = 3 [ sp2 hybridization ]
NH4+ :
The central atom is Nitrogen
The electronic configuration of Nitrogen is 1s22s22p3
The valence electrons present in nitrogen is 5
- The hybridization of nitrogen in NH4+
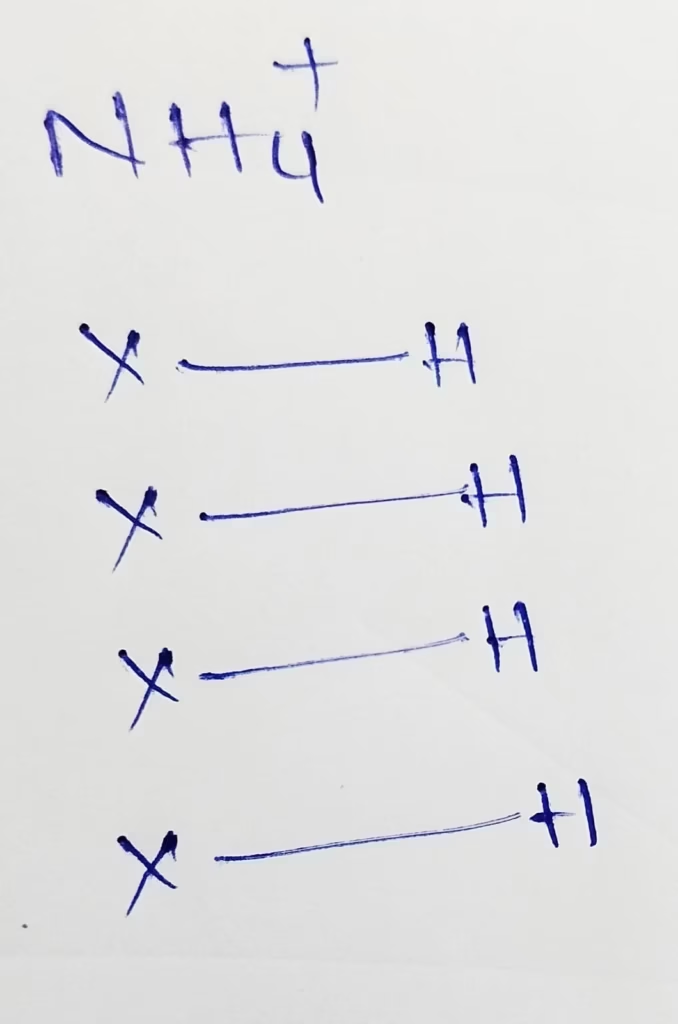
Hybridization : Number of bonds + Lone pair = 4 + 0 lone pair = 4 [sp3 hybridization]
Correct answer : Option (a)
2. Dipole moment of NF3 is smaller than :
(a) NH3
(b) CO2
(c) BF3
(d) CCl4
Answer :
Correct answer is Option (a)
NH3 :
The central atom is Nitrogen
The electronic configuration of Nitrogen is 1s22s22p3
The valence electrons present in nitrogen is 5
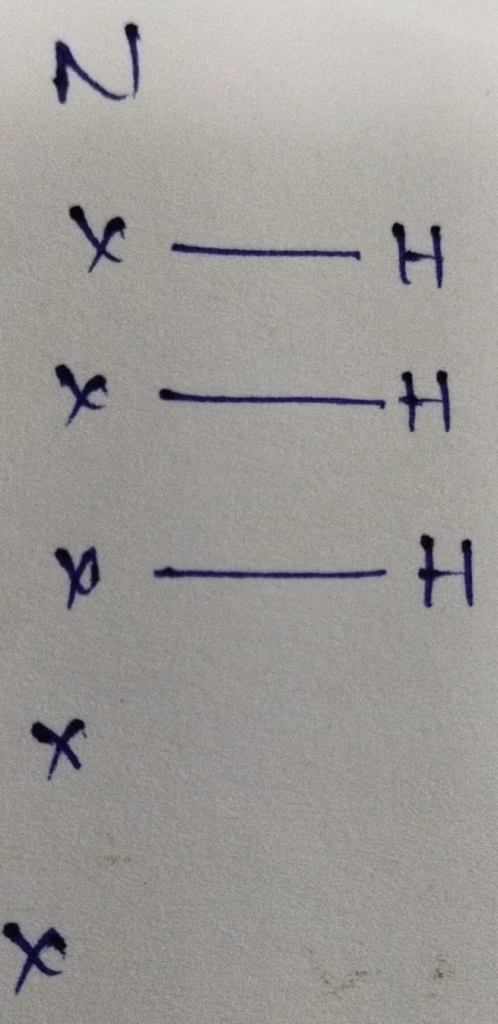
Hybridization = Number of bonds + Lone pair = 3 bonds + 1 lone pair = 4 [ sp3 hybridization ]
The shape of NH3 molecule is pyramidal
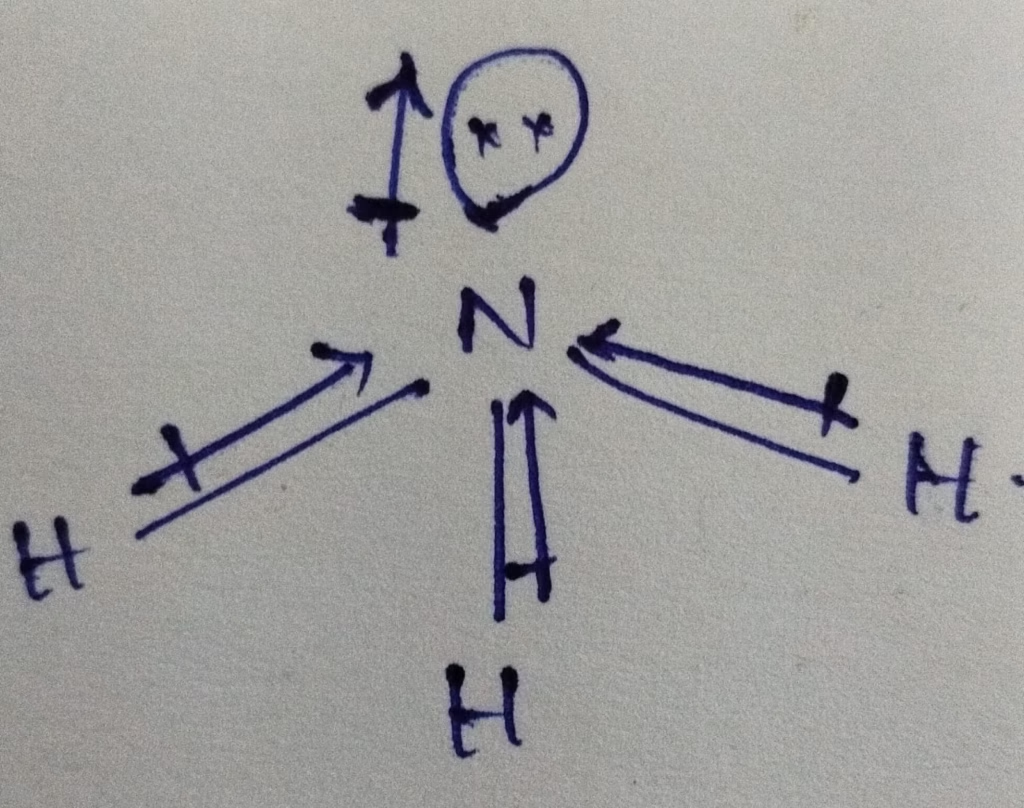
Here, N—H dipole moment and N – orbital lone pair dipole moment in same direction so the dipole moment NF3 is smaller than NH3
